Lipostatic hypothesis: Difference between revisions
imported>Sophie Rajska |
imported>Sophie Rajska |
||
| Line 50: | Line 50: | ||
Wade, G. N. (2004). "Regulation of body fat content?" American Journal of Physiology - Regulatory, Integrative and Comparative Physiology 286(1): R14-R15. | Wade, G. N. (2004). "Regulation of body fat content?" American Journal of Physiology - Regulatory, Integrative and Comparative Physiology 286(1): R14-R15. | ||
Zhang, Y., R. Proenca, et al. (1994). "Positional cloning of the mouse obese gene and its human homologue." Nature 372(6505): 425-432. | |||
Zhang Y & Scarpace PJ (2006) The role of leptin in leptin resistance and obesity "Physiol Behav" 88:249-256. | Zhang Y & Scarpace PJ (2006) The role of leptin in leptin resistance and obesity "Physiol Behav" 88:249-256. | ||
Revision as of 07:45, 25 October 2011
For the course duration, the article is closed to outside editing. Of course you can always leave comments on the discussion page. The anticipated date of course completion is 01 April 2012. One month after that date at the latest, this notice shall be removed. Besides, many other Citizendium articles welcome your collaboration! |
In 1953, Kennedy [1] proposed what became known as the lipostatic hyothesis. Specifically, he postulated the existence, in the hypothalamus, of a centre that was sensitive to the concentration of metabolites in the circulation, which prevented "an overall surplus of energy intake over expenditure."
Introduction
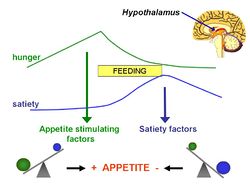
The lipostatic hypothesis describes the feedback mechanisms between adipose tissue deposition and hypothalamic signalling, in order to sustain 'lipostasis'. Lipostasis is achieved through the modulation and balancing of energy intake and expenditure to prevent an individual 'getting fat'. For the brain to know when their is too much adipose tissue building up, a signalling hormone must be released from the adipose tissue to signal to the brain that a satiation signal must be released in order to stop the individual from eating. In 1994, it was discovered that this hormone released from adipose tissue, and critical in regulating the size of the body fat depot, was coded for by the ob gene and is known as leptin[2]
What is leptin?
Leptin is a adipocite derived hormone that circulates in the blood in proportion to whole body adipose tissue mass. In other words, except for ob/ob individuals, the more adipose tissue an individual contains, the greater the amount of leptin they have circulating in their blood. Leptin is a hydrophobic hormone and can therefore cross the blood-brain barrier and act on specific leptin receptors that are located in the ventromedial hypothalamic nucleus (VMN). The leptin signal transmits information about the size of body fat depots, binds to leptin receptors in the VMN, causing the production of POMC, which causes satiation.
Leptin was discovered as a product of the ob gene and its role in regulating food intake was identified through ob/ob knockout mice. These ob deficient mice could not produce leptin and therefore had no means of starting the feedback mechanism, as there was no signalling to the VMN of excess adipose tissue. Due to this, there is no anorexigenic response and the ob/ob mice keeps on eating due to a lack of satiation feeling. Another mutation that further supported leptins role in energy balance came from db/db deficient mice. These mice expressed leptin insensitive receptors and therefore, even though their adipose tissue produced leptin in abundance, the lack of leptin sensitive receptors also meant that there was no complete signal to the brain of excess adipose tissue.
Interestingly, leptin treatment of these two different mutant mice led to drastically different results. The ob/ob mice, when treated with leptin over time, lost a vast amount of weight as leptin elicits the anorexigenic response that the mouse could not provide before. The db/db mice however had no weight reduction from the introduction of leptin, as their receptors were still insensitive to the hormone.
Hypothalamic leptin signaling
Leptin receptor mediated hypothalamic signaling involves multiple pathways. The JAK2/cytosolic signal transducer and activator of transcription protein 3 (STAT3) pathway plays an important role, although the JAK2-Phosphotidylinosital 3 kinase (PI3K) pathway has also shown to be important. With respect to the JAK2/STAT3 pathway, leptin binding to the long form of the leptin receptor (Ob-Rb) initiates tyrosine phosphorylation of Ob-Rb by JAK2. Phosphorylated leptin receptor then recruits STAT3 that is activated through phosphorylation by JAK2. The activated STAT3 proteins dimerize and translocate to the nucleus where they bind DNA and initiate gene transcription. Suppresser of cytokine signaling 3 (SOCS-3) and phosphotyrosine phosphotase 1B (PTP1B) are two known negative regulators of leptin signaling following Ob-Rb activation. The JAK2/STAT3 pathway is believed to be essential for mediating leptin's effects on homeostatic energy regulation.
Parabolis evidence
Conclusion
References
Cowley M et al. (2001) Leptin activates anorexigenic POMC neurones through a neutral network in the arcuate nucleus 411: 480-484
Crowley V et al. (2002) Obesity therapy: Altering the Energy Intake-And-Expenditure Balance Sheet 1: 276-284
López, M., C. J. Lelliott, et al. (2007). "Hypothalamic fatty acid metabolism: A housekeeping pathway that regulates food intake." BioEssays 29(3): 248-261.
Romanovsky, A. A. (2007). "Thermoregulation: some concepts have changed. Functional architecture of the thermoregulatory system." American Journal of Physiology - Regulatory, Integrative and Comparative Physiology 292(1): R37-R46.
Wade, G. N. (2004). "Regulation of body fat content?" American Journal of Physiology - Regulatory, Integrative and Comparative Physiology 286(1): R14-R15.
Zhang, Y., R. Proenca, et al. (1994). "Positional cloning of the mouse obese gene and its human homologue." Nature 372(6505): 425-432.
Zhang Y & Scarpace PJ (2006) The role of leptin in leptin resistance and obesity "Physiol Behav" 88:249-256.