Volatility (chemistry): Difference between revisions
imported>Peter Schmitt (→Relative volatility: separation of two components needs higher relative volatility of these two components) |
imported>Milton Beychok m (→Relative volatility: Peter, I agree it looks better in just plain HTML ... and it looks even better in bold font. Thanks.) |
||
| Line 21: | Line 21: | ||
{{main|Relative volatility}} | {{main|Relative volatility}} | ||
''[[Relative volatility]]'' is a measure of the difference between the vapor pressure of the more volatile components of a liquid mixture and the vapor pressure of the less volatile components of the mixture. This measure is widely used in designing large industrial [[Continuous distillation|distillation]] processes.<ref name=Kister/><ref name=Perry/><ref name=Seader>{{cite book|author= Seader, J. D., and Henley, Ernest J.|title=Separation Process Principles|publisher=Wiley| location=New York|year=1998|id=ISBN 0-471-58626-9}}</ref> In effect, it indicates the ease or difficulty of using distillation to separate the more volatile components from the less volatile components in a mixture. In other words, the higher is the relative volatility (of two components) of a liquid mixture, the easier it is to separate these two components by distillation. By convention, relative volatility is typically denoted as α. | ''[[Relative volatility]]'' is a measure of the difference between the vapor pressure of the more volatile components of a liquid mixture and the vapor pressure of the less volatile components of the mixture. This measure is widely used in designing large industrial [[Continuous distillation|distillation]] processes.<ref name=Kister/><ref name=Perry/><ref name=Seader>{{cite book|author= Seader, J. D., and Henley, Ernest J.|title=Separation Process Principles|publisher=Wiley| location=New York|year=1998|id=ISBN 0-471-58626-9}}</ref> In effect, it indicates the ease or difficulty of using distillation to separate the more volatile components from the less volatile components in a mixture. In other words, the higher is the relative volatility (of two components) of a liquid mixture, the easier it is to separate these two components by distillation. By convention, relative volatility is typically denoted as '''α'''. | ||
== Volatile Organic Compound == | == Volatile Organic Compound == | ||
Revision as of 16:38, 22 September 2010
In chemistry and physics, volatility is a term used to characterize the tendency of a substance to vaporize.[1] It is directly related to a substance' s vapor pressure. At a given temperature, a substance with a higher vapor pressure will vaporize more readily than a vapor with a lower vapor pressure.[2][3][4]
In common usage, the term applies primarily to liquids. However, it may also be used to characterize the process of sublimation by which certain solid substances such as ammonium chloride (NH4Cl) and dry ice, which is solid carbon dioxide (CO2), change directly from their solid form to a vapor without becoming a liquid.
Any substance with a significant vapor pressure at temperatures of about 20 to 25 °C (68 to 77 °F) is very often referred to as being volatile.
Vapor Pressure, temperature and boiling point
The vapor pressure of a substance is the pressure at which its gaseous (vapor) phase is in equilibrium with its liquid or solid phase. It is a measure of the tendency of molecules and atoms to escape from a liquid or solid. A liquid's boiling point at atmospheric pressure corresponds to the temperature at which its vapor pressure is equal to the surrounding atmospheric pressure and is very commonly referred to as the normal boiling point.
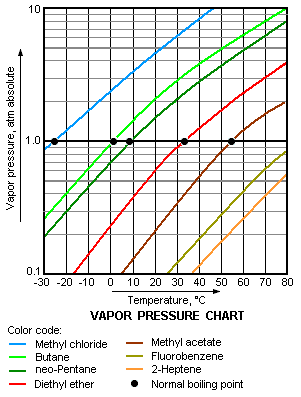
The higher is the vapor pressure of a liquid, the higher is the volatility and the lower is the normal boiling point of the liquid. The adjacent vapor pressure chart graphs the dependency of vapor pressure upon temperature for a variety of liquids[5] and also confirms that liquids with higher vapor pressures have lower normal boiling points.
For example, at any given temperature, methyl chloride (CH3Cl) has the highest vapor pressure of any of the liquids graphed in the chart. It also has the lowest normal boiling point (−26 °C), which is where its vapor pressure curve (the blue line) intersects the horizontal pressure line of one atmosphere (atm) of absolute vapor pressure.
In terms of intermolecular forces, the boiling point represents the point at which the liquid molecules possess enough thermal energy to overcome the various intermolecular attractions binding the molecules into the liquid. Therefore the boiling point is also an indicator of the strength of those attractive forces. The higher the intermolecular attractive forces are, the more difficult it is for molecules to escape from the liquid and hence the lower is the vapor pressure of the liquid. The lower the vapor pressure of the liquid, the higher the temperature must be to initiate boiling. Thus, the higher the intermolecular attractive forces are, the higher is the normal boiling point.[6]
Relative volatility
Relative volatility is a measure of the difference between the vapor pressure of the more volatile components of a liquid mixture and the vapor pressure of the less volatile components of the mixture. This measure is widely used in designing large industrial distillation processes.[4][5][7] In effect, it indicates the ease or difficulty of using distillation to separate the more volatile components from the less volatile components in a mixture. In other words, the higher is the relative volatility (of two components) of a liquid mixture, the easier it is to separate these two components by distillation. By convention, relative volatility is typically denoted as α.
Volatile Organic Compound
Volatile organic compound (VOC) is a term that refers to organic chemical compounds having significant vapor pressures and which can have adverse effects on the environment and human health. VOCs are numerous, varied and include man-made (anthropogenic) as well as naturally occurring chemical compounds. The anthropogenic VOCs are regulated by various governmental environmental entities worldwide.
There is no universally accepted definition of VOCs. Some regulatory entities define them in terms of their vapor pressure at ordinary temperatures, or their normal boiling points, or how many carbon atoms they contain per molecule, and others define them in terms of their photochemical reactivity.
The U.S. Environmental Protection Agency currently defines them as any compound of carbon, excluding carbon monoxide, carbon dioxide, carbonic acid, metallic carbides or carbonates, and ammonium carbonate, which participates in atmospheric photochemical reactions (i.e., the reactions that produce photochemical smog. However, any such carbon compounds that have been determined to have a low photochemical reactivity, and specifically listed in the regulation, are exempted from regulation.[8]
References
- ↑ Note: To vaporize means to become a vapor.
- ↑ Gases and Vapor (University of Kentucky website)
- ↑ James G. Speight (2006). The Chemistry and Technology of Petroleum, 4th Edition. CRC Press. ISBN 0-8493-9067-2.
- ↑ 4.0 4.1 Kister, Henry Z. (1992). Distillation Design, 1st Edition. McGraw-Hill. ISBN 0-07-034909-6.
- ↑ 5.0 5.1 R.H. Perry and D.W. Green (Editors) (1997). Perry's Chemical Engineers' Handbook, 7th Edition. McGraw-Hill. ISBN 0-07-049842-5.
- ↑ The Nature of Intermolecular Forces From the Louisiana State University Chemistry Department website.
- ↑ Seader, J. D., and Henley, Ernest J. (1998). Separation Process Principles. New York: Wiley. ISBN 0-471-58626-9.
- ↑ U.S. Code of Federal Regulations: 40 CFR 51.100(s) - Definition - Volatile organic compounds (VOC)
- Pages using ISBN magic links
- CZ Live
- Chemistry Workgroup
- Biology Workgroup
- Engineering Workgroup
- Chemical Engineering Subgroup
- Environmental Engineering Subgroup
- Articles written in American English
- Advanced Articles written in American English
- All Content
- Chemistry Content
- Biology Content
- Engineering Content
- Chemical Engineering tag
- Environmental Engineering tag